Dec, 2021
Presented by Malini Veerappan Pasricha, MD


Presented by Malini Veerappan Pasricha, MD

An asymptomatic 33-year-old Middle Eastern male was referred for presumed central serous chorioretinopathy of the right eye
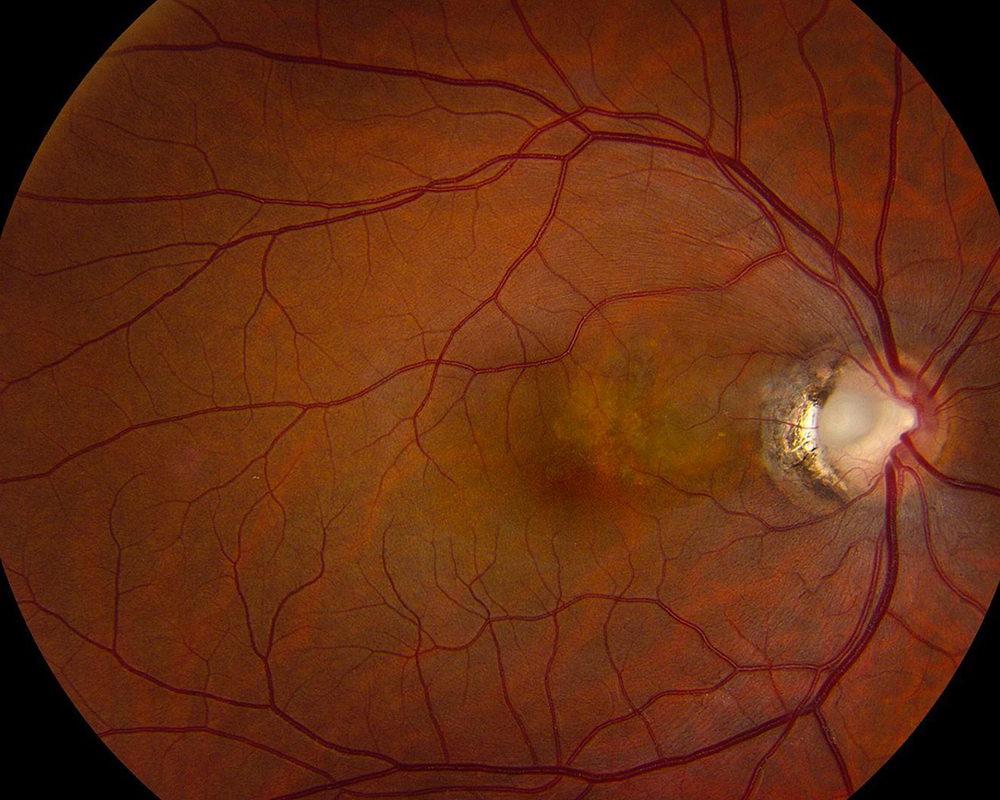
Figure 1: Fundus color photograph of the right eye. Note the gray depression at the temporal margin of the optic nerve head, with associated temporal peripapillary atrophy. There is also retinal pigment epithelial changes and lipofuscin accumulation superonasal to the fovea.
The patient denied any changes in visual acuity, metamorphopsia, or headaches. He was a contact lens wearer for moderate myopia, with no other past ocular history. His past medical history was unremarkable. Family history was non-contributory. He was not taking any medications, including oral or inhaled corticosteroids. He denied use of alcohol, cigarettes, or illicit substances. Review of systems was negative.
His best corrected Snellen visual acuity measured 20/16 in both eyes (OU). His intraocular pressure was 14 mmHg OU. Anterior segment examination was unremarkable in both eyes. Fundus exam of the right eye showed a gray excavation of the temporal optic nerve, temporal peripapillary chorioretinal atrophy and a patch of retinal pigment epithelial changes superonasal to the fovea (Figure 1). Fundus exam of the left eye was unremarkable. Spectral domain optical coherence tomography (SD-OCT) through the fovea showed normal foveal contour in both eyes, with trace extra-foveal subretinal fluid and trace peripapillary macular edema in the right eye (Figure 2). SDOCT of the papillary region showed optic disc excavation with peripapillary subretinal fluid, consistent with a localized and fovea-sparing serous retinal detachment (Figures 3A and 3B). Fundus autofluoroscence of the right eye showed hyper-autofluorescence at the edge of the pocket of subretinal fluid, corresponding to lipofuscin accumulation (Figure 4). Late phase fluorescein angiogram showed mild hyper-fluorescence superonasal to the fovea corresponding to the subretinal fluid (Figure 5).
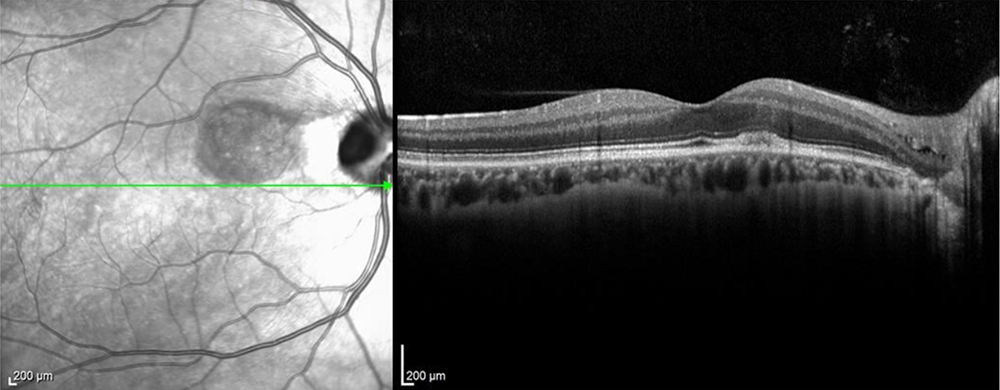
Figure 2: SD-OCT of the right eye. A horizontal scan through the fovea shows a normal foveal contour, though trace extra-foveal subretinal fluid and trace peripapillary macular edema can be noted.
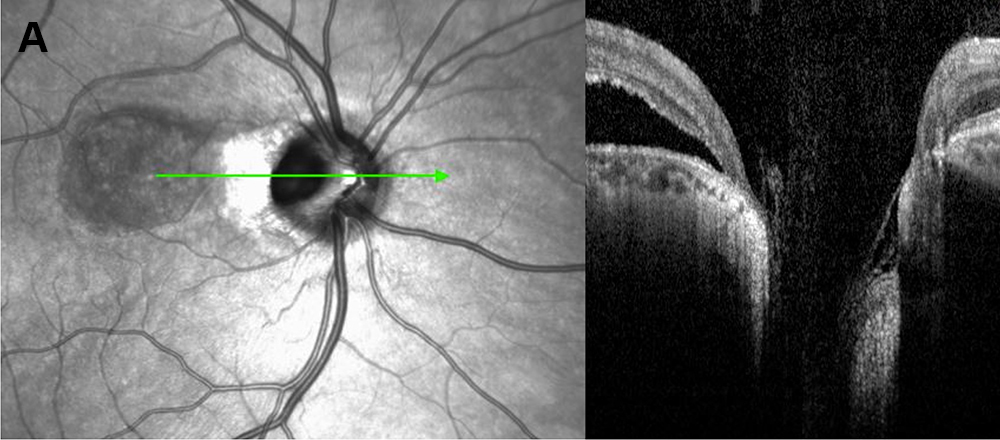
Figure 3A: SD-OCT of the right eye. A horizontal scan through the optic nerve shows an optic disc pit with peripapillary subretinal fluid.
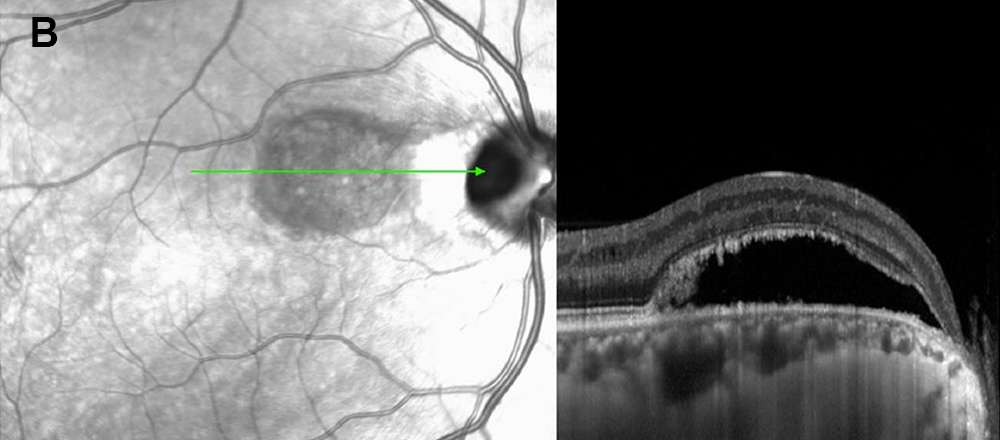
Figure 3B: SD-OCT of the right eye. A horizontal scan through the papillomacular bundle shows RPE changes adjacent to the optic nerve and serous retinal detachment with shaggy photoreceptors.
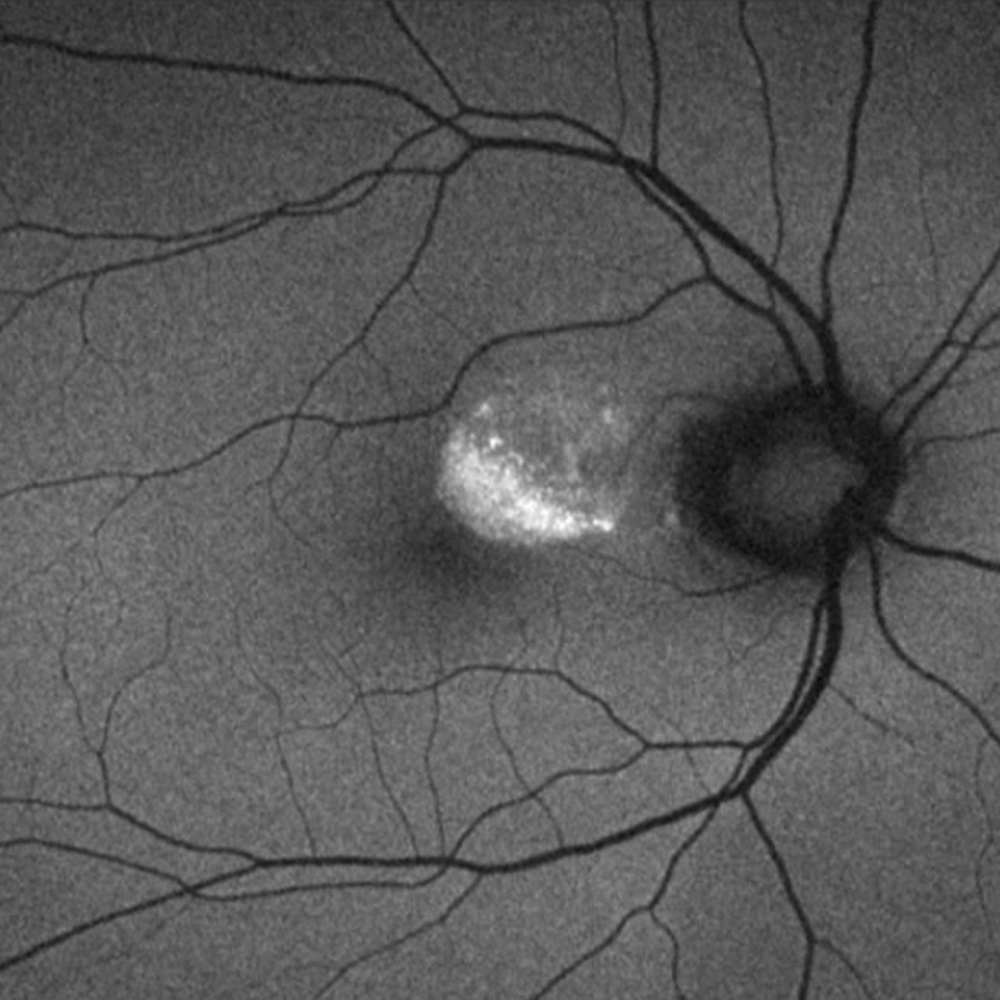
Figure 4: Fundus autofluoroscence of the right eye. Note the hyper-autofluorescence at the edge of the pocket of subretinal fluid, corresponding to lipofuscin accumulation.
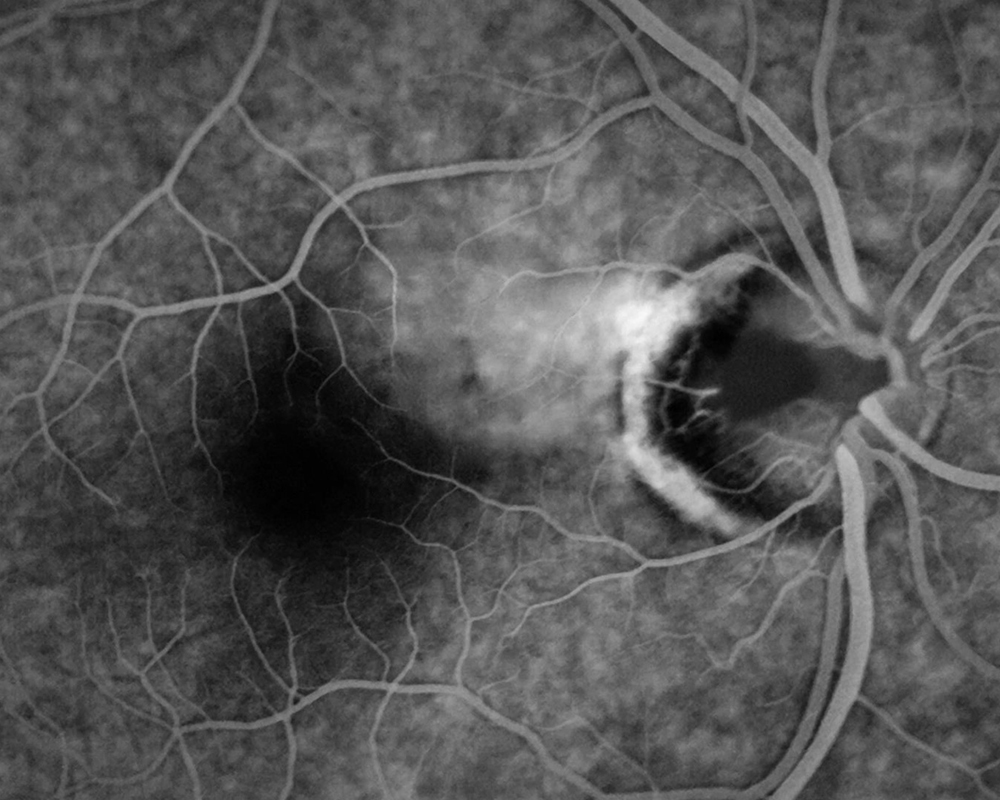
Figure 5: Late phase fluorescein angiogram of the right eye. Note the irregular hyperfluorescence in the PMB corresponding to RPE changes as well as mild leakage. The optic pit shows absent fluorescence.
Discussion
Congenital optic pit is a rare (1 out of 10,000 people) anomaly of the optic nerve head that is presumed to result from incomplete closure of the embryonic fissure. It is bilateral in 10-15% of cases, and men and women are equally affected. Most cases are nonfamilial, though there have been a few reports of an autosomal dominant pattern of inheritance.1,2
An optic pit appears as a dark gray depression in the optic nerve head, although it can vary from black to yellow in color.1 Size is variable, with the normal range being between 1/8 and 1/4 of the disc area.1 About 70% are located on the temporal margin of the disc, while another 20% are located centrally.1 Histopathology has shown the optic pit to contain dysplastic retina that extends through a defect in the lamina cribrosa into the subarachnoid space.2
Serous retinal detachments are known to be a common complication of optic disc pits, occurring in 40-50% of patients3 (though the true percentage may be higher given that patients with fovea-sparing macular detachments may be asymptomatic and therefore not present for evaluation). The complication occurs most frequently with optic pits that are larger in size and those located in the temporal disc.1 In fact, some cases of optic pit-related serous retinal detachments have a conduit visible on OCT between the temporal optic pit and pocket of intraretinal or subretinal fluid.1
There are several theories about the pathophysiology of serous macular detachments, and most are based on OCT findings. The most generally accepted theory is that fluid, from either the vitreous cavity or the subarachnoid space, enters through the optic pit and travels between the inner and outer layers of the retina to produce retinoschisis.4 Fluid can also enter the subretinal space either by direct flow through the optic pit, or due to a hole in the outer retina creating a communication between the retinoschisis and subretinal space (Figures 6A and 6B).1,2
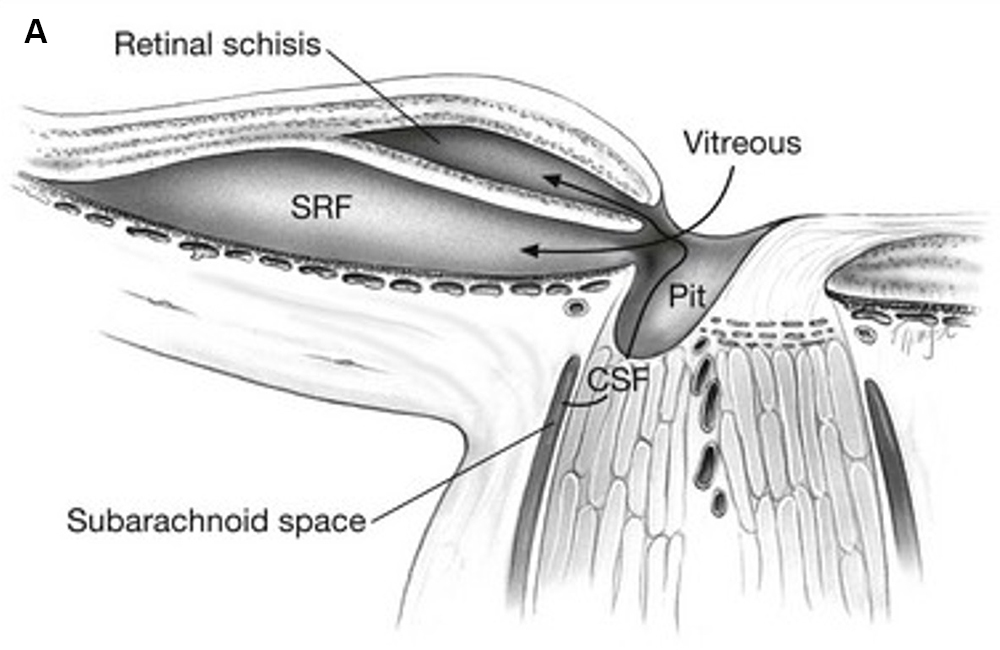
Figure 6A: Schematic drawing of axial view of optic nerve head, showing mechanisms by which fluid enter the intraretinal and subretinal space. Fluid from the vitreous cavity or subarachnoid space enters the intraretinal and subretinal spaces independently. Source: Entokey.com
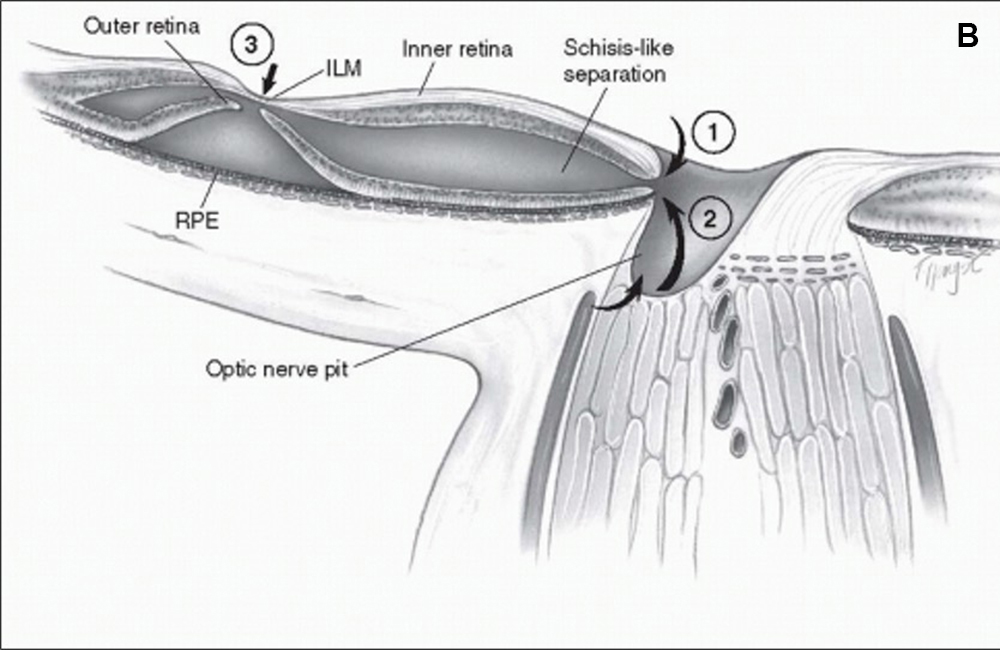
Figure 6B: Fluid from the vitreous cavity or subarachnoid space enters the intraretinal space, and a break in the outer retinal layer subsequently allows entry of fluid into the subretinal space. Source: Entokey.com
Optic pits are most commonly associated with two types of visual field defects: arcuate scotomas due to absence of nerve fiber layers displaced by the optic disc pit, and central scotomas due to serous macular detachments.1 The prognosis for return of vision after a serous macular detachment is variable and dependent on chronicity of detachment.5 For fovea-involving macular detachments, 80% of untreated patients lost vision to 20/200 or worse.5 Vision loss is generally complete within 6 months and long term visual outcomes have been correlated with initial presenting visual acuity.5 Long term macular changes include “upside down” lamellar retinal holes (ILM is generally spared), full thickness macular holes, RPE mottling, and chronic cystic changes of the macula.5
Due to the rare nature of optic disc pits, most studies evaluating treatment options are small and nonrandomized, leading to difficulty in coming to a consensus on best management strategies.6,7 Some studies demonstrate the role of vitreous traction in the pathophysiology of optic disc maculopathy,8,9 and thus advocate for vitrectomy with induction of posterior vitreous detachment to release traction and aid in re-attachment of the retina.10-12 Another proposed technique is in-office gas fluid exchange, with prone positioning. Gas tamponade is thought to be important in moving the subretinal fluid away from the macula and spreading it over a greater area of RPE, thereby enhancing fluid resorption.13 Another strategy involves laser photocoagulation, applied in rows between the retinal detachment and optic disc, to diminish inflow of fluid into the schisis cavity or subretinal space.3,14 Other strategies with variable results are systemic steroids, optic nerve sheath decompression, scleral buckling, peeling of the internal limiting membrane and partial or full thickness drainage retinotomies.15-18 Combinations of the above techniques have also been employed.19,20 No technique has proven superior and often multiple treatments are needed to achieve retinal reattachment. Furthermore, while many of these techniques have been shown to achieve anatomic success in re-attaching the retina, their role in improving visual acuity is questionable.
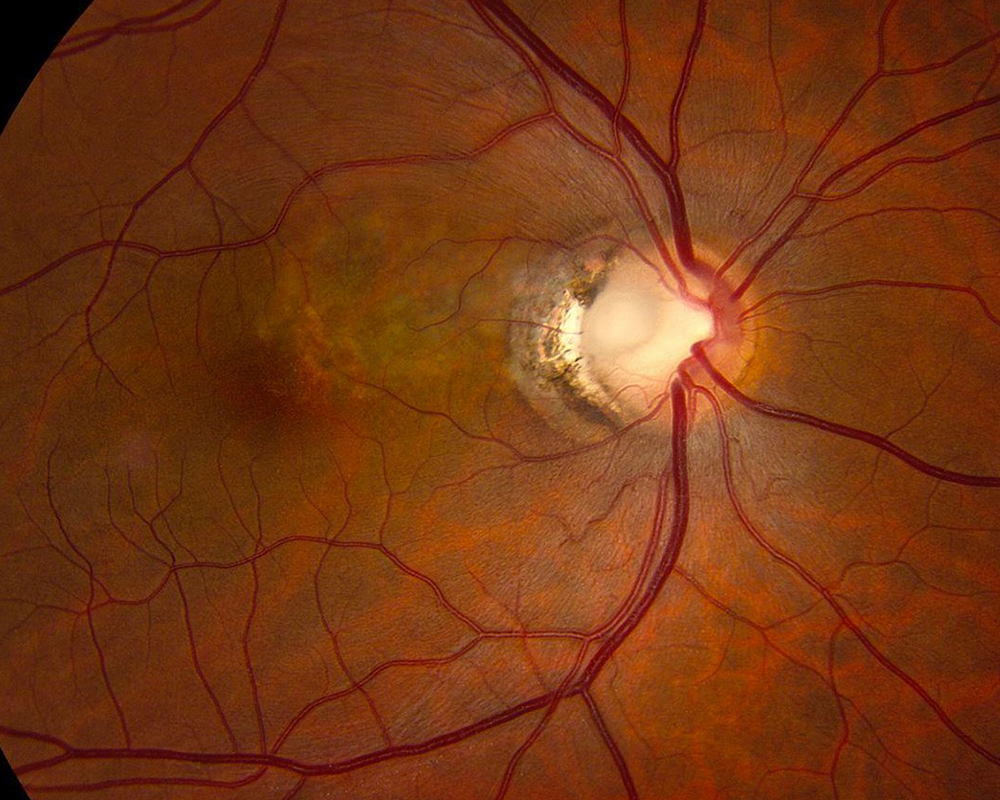
Figure 7: Fundus photo of the right eye 3 years after presentation. No treatment was undertaken and the subretinal has not progressed.
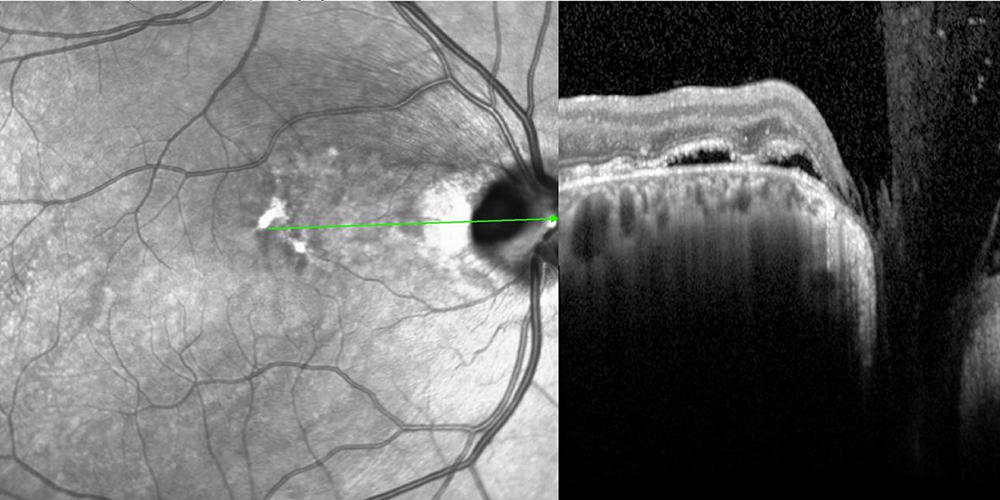
Figure 8: SD-OCT of the right eye 3 years after presentation. No treatment was undertaken and the subretinal has not progressed.
The natural history of fovea-sparing macular detachments are not as well studied due to ascertainment bias. Given excellent visual acuity and lack of foveal involvement in our patient, the decision was made to observe closely with serial exams and imaging. The patient was advised to self-monitor any visual changes using an Amsler grid. On most recent evaluation – three years after his initial presentation and with no intervention– the subretinal fluid has remained stable and the fovea has remained attached with maintenance of best corrected visual acuity at 20/16 (Figures 7 and 8).