March, 2021
Presented by Caleb Ng, MD


Presented by Caleb Ng, MD

A 68-year-old Caucasian man presented with chief complaint of decreased vision and floaters affecting both eyes.
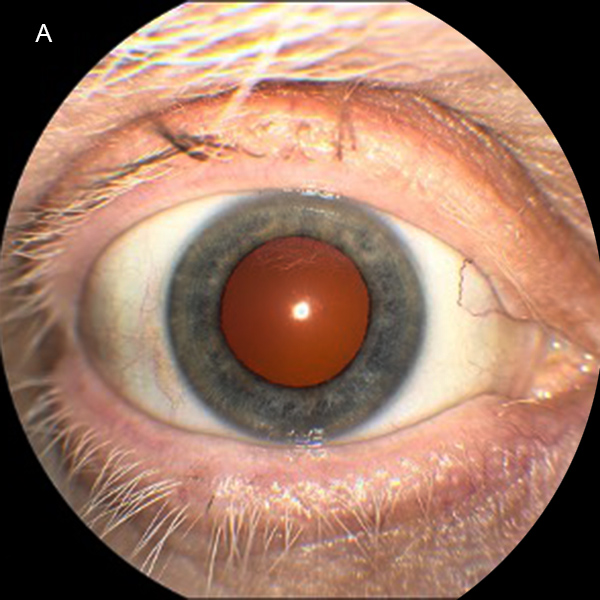
Figure 1A: Color photograph showing poliosis of the patient's eyelashes.
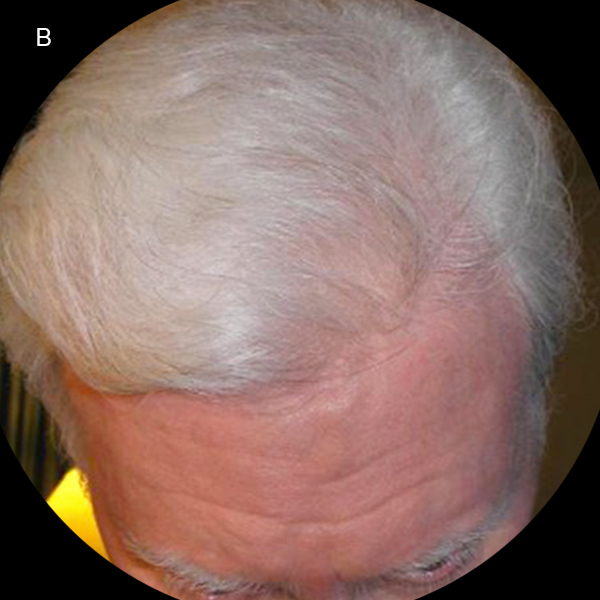
Figure 1B: Color photograph showing diffuse poliosis of patient's hair and eyebrows.
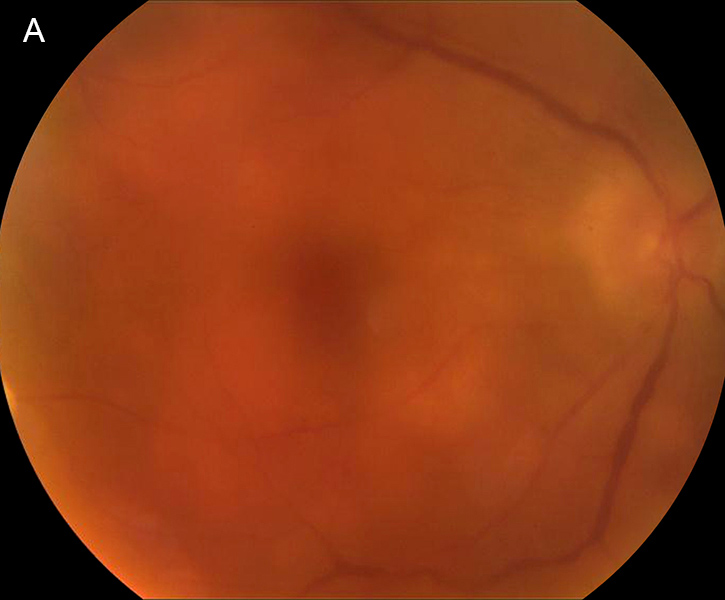
Figure 2A: Color photograph of the right eye. Note the marked media haze that was secondary to vitritis. Optic nerve edema is present, and multiple deep yellowish lesions throughout the macula.
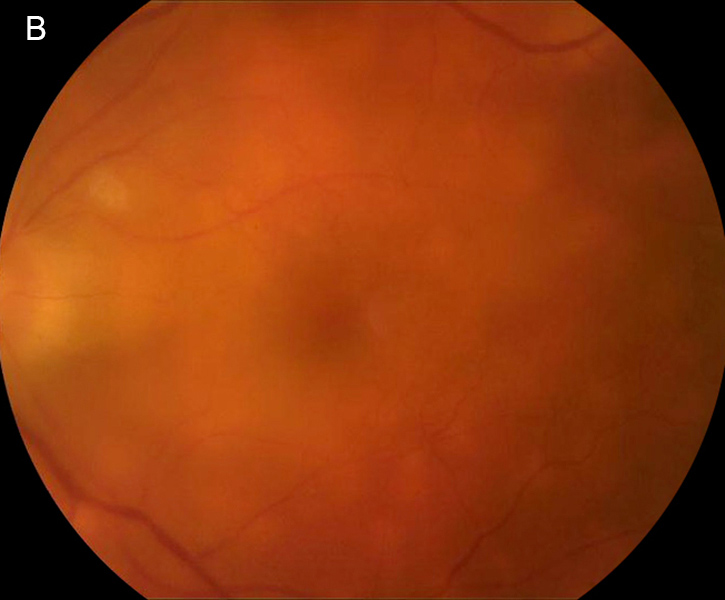
Figure 2B: Color photograph of the left eye. Similar findings are present as were seen in the left eye. There is marked media haze that was secondary to vitritis, optic nerve edema, and multiple deep yellowish lesions throughout the macula.
The patient’s best-corrected visual acuity (BCVA) was 20/60 in the right eye and 20/80 in the left. His external appearance was remarkable for diffuse poliosis of the scalp, eyebrows, and eye lashes, and vitiligo of the forehead skin by the hair line (Figures 1A and B). Examination of the anterior segments revealed small keratic precipitates, mild anterior chamber inflammation, and trace anterior vitreous cell in both eyes. Posterior segment examination revealed bilateral findings of moderate vitreous haze, serous macular detachments, and numerous multifocal deep yellow-orange lesions in the posterior poles (Figures 2A and 2B). Fluorescein angiography revealed disc leakage and diffuse staining of the large retinal vessels, without retinal pigment epithelium (RPE) leaks, choroidal neovascularization (CNV), or cystoid macular edema (Figures 3A and 3B). Indocyanine green angiography showed numerous hypofluorescent spots in both eyes (Figures 4A and 4B). Optical coherence tomography (OCT) showed bilateral serous retinal detachments involving the central macula of both eyes (Figures 5A and 5B).
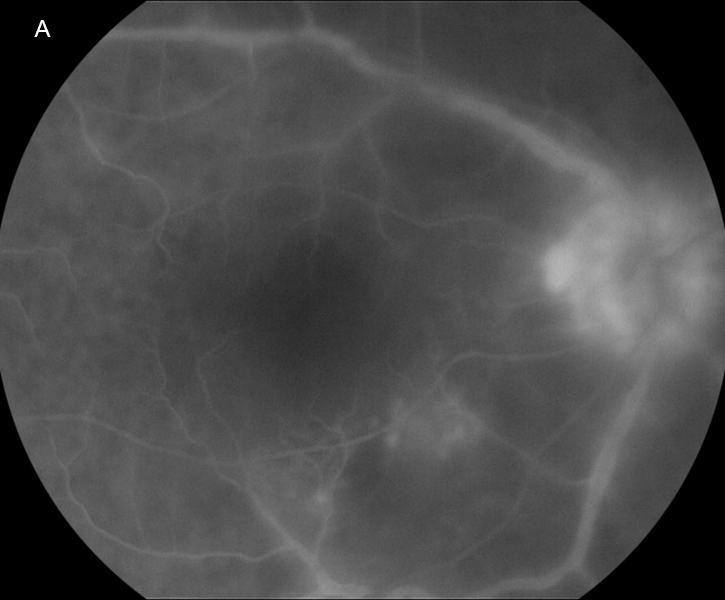
Figure 3A: Fluorescein angiogram of the right eye. Note the significant vascular and disc leakage. There are no puntate areas of RPE leakage.
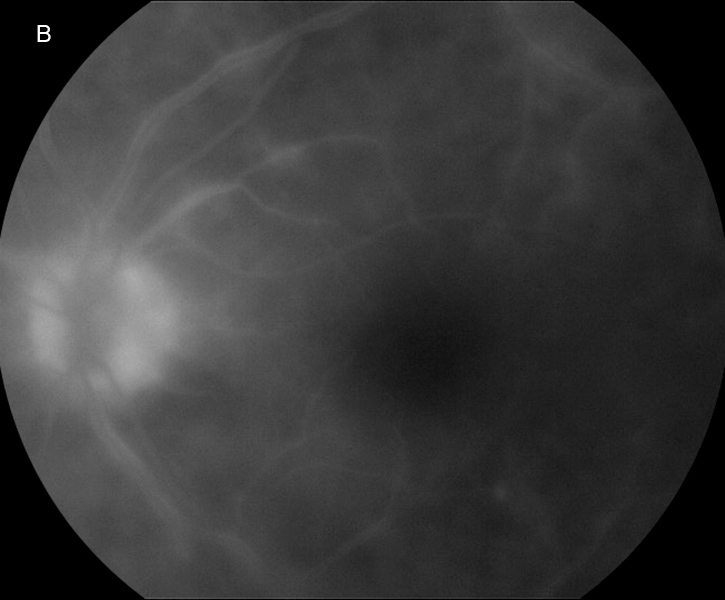
Figure 3B: Fluorescein angiogram of the left eye. Angiographic findings are similar to what was present in the right eye.
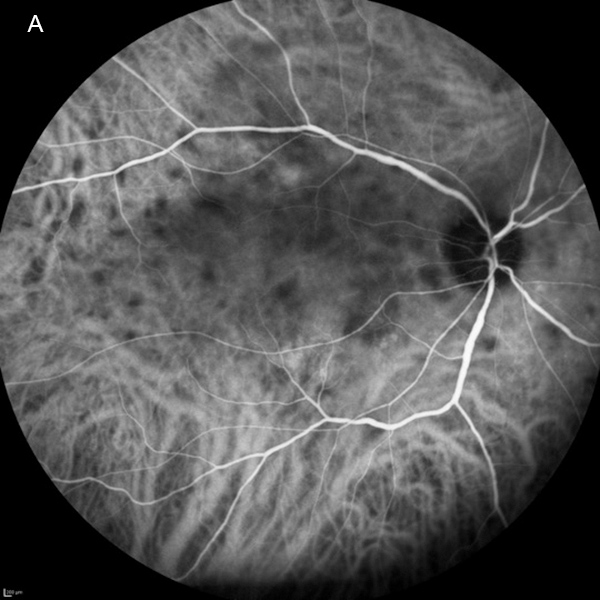
Figure 4A: Indocyanine green angiogram of the right eye. Note the multiple areas of hypofluorescent areas scattered throughout the posterior pole.
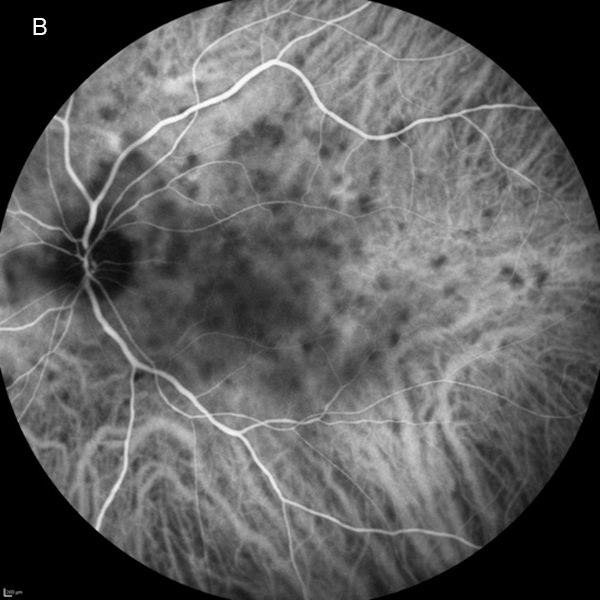
Figure 4B: Indocyanine green angiogram of the left eye. Note the multiple areas of hypofluorescent areas scattered throughout the posterior pole.
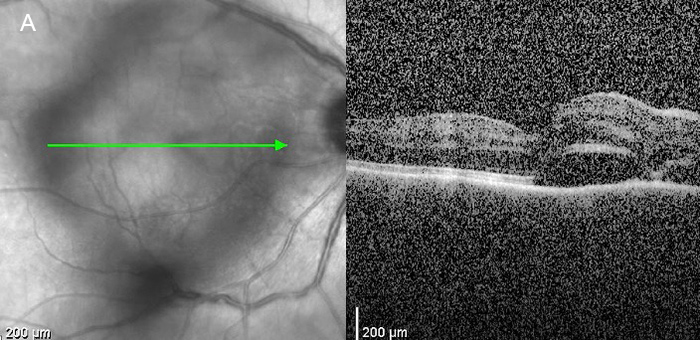
Figure 5A: OCT scan of the right macula. Note the serous retinal detachment.
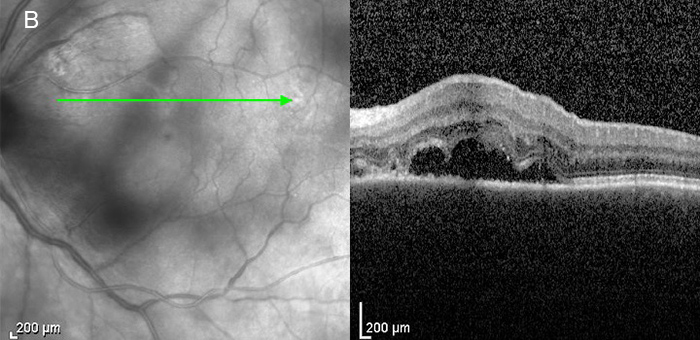
Figure 5B: OCT scan of the left macula. Note the serous retinal detachment.
Past medical history was notable for cutaneous melanoma of the right ankle diagnosed one year prior to presentation, and treated with surgical excision, including negative regional lymph node biopsies. A review of systems was notable for patchy loss of scalp hair over the prior two months, and the occurrence of a diffuse skin rash, three months prior to presentation, which on biopsy was found to be consistent with dermatomyositis.
Differential Diagnosis
Work-Up and Diagnosis
Purified protein derivative testing was negative. Serologic evaluation that included a complete blood count, angiotensin converting enzyme and lysozyme levels, fluorescent treponemal antibody titers, a rapid plasma reagin test, antineutrophil cytoplasmic antibodies, erythrocyte sedimentation rate, C-reactive protein, and serologic testing for exposure to Hepatitis B and C, human immunodeficiency virus, and Borrelia burgdorferi were all normal or negative. Chest x-ray and magnetic resonance imaging of the brain were unremarkable, but a whole body computed tomography (CT) and positron emission tomography (PET) scan detected a pelvic mass. Surgical excision and histologic examination of the mass and its adjacent lymph nodes confirmed the diagnosis of metastatic melanoma, but no ensuing treatment with chemotherapy, radiation, or checkpoint inhibitor class medications was initiated. The patient was diagnosed with paraneoplastic VKH-like uveitis in the setting of metastatic cutaneous melanoma. He was treated with prednisone and given multiple posterior sub-Tenon’s corticosteroid injections bilaterally with eventual resolution of inflammation and disappearance of the multifocal yellow-orange lesions (Figures 6A and 6B). The patient’s BCVA recovered to 20/20 OD and 20/25 OS one year following his initial presentation, and there has been no recurrence of the VKH-like uveitis.
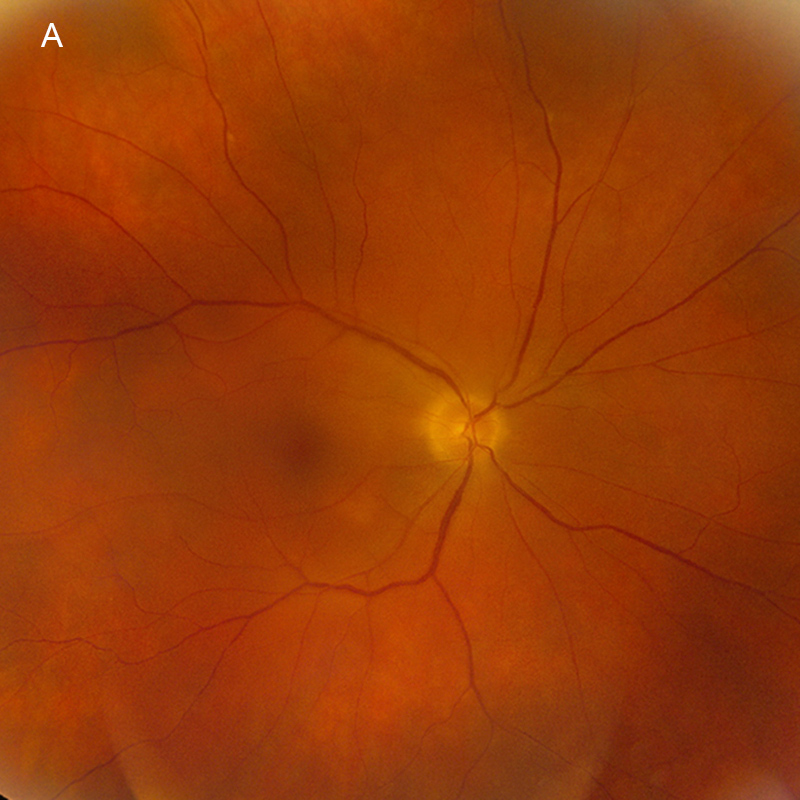
Figure 6A: Color photograph of the right eye. Note the marked improvement in the media clarity and resolution of the optic nerve edema and deep yellowish lesions.
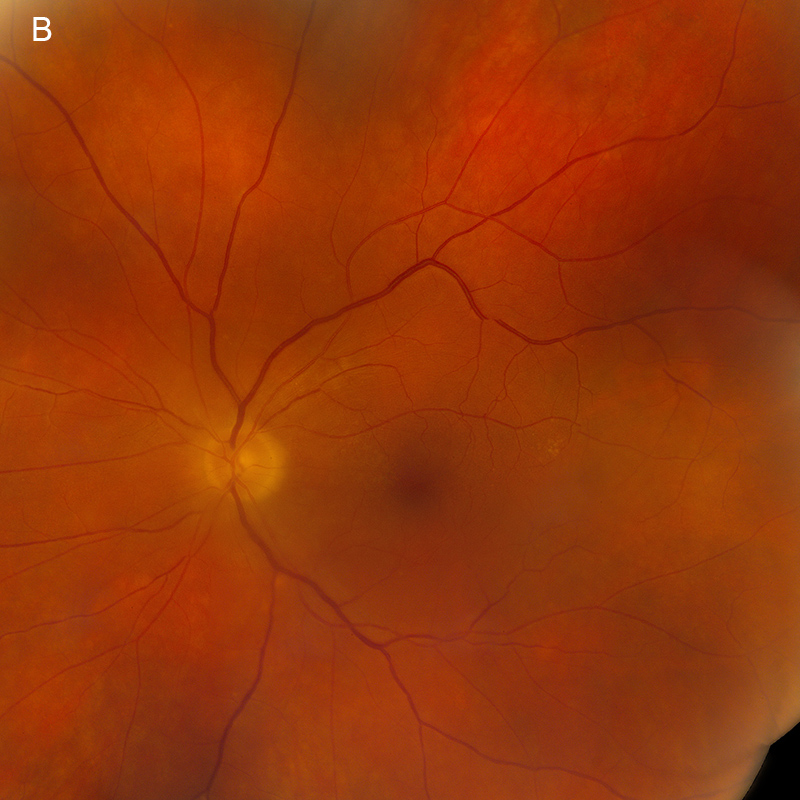
Figure 6A: Color photograph of the left eye. Note the marked improvement in the media clarity and resolution of the optic nerve edema and deep yellowish lesions.
Additional History
This patient returned two years later for evaluation of progressive nyctalopia. The BCVA had reduced to 20/50 in the right eye and 20/40 in the left eye. Examination of both the anterior and posterior segments showed no intraocular inflammation, but new focal areas of chorioretinal atrophy were noted in each eye (images not shown). Fluorescein angiography revealed hyperfluorescent window defects in areas of RPE atrophy in both eyes (images not shown). Humphrey visual field testing showed central and peripheral visual field loss in the right eye, and a dense ring scotoma in the left eye (images not shown). Full-field electroretinography (ERG) to dim flash was nondetectable and electronegative tracing to standard flash (Figure 7). A serum autoimmune retinopathy panel detected antibodies against enolase, arrestin, and pyruvate kinase M2 which, in combination with the results of functional testing, were felt to be consistent with a diagnosis of MAR. A repeat PET-CT scan showed no evidence of additional metastatic disease.
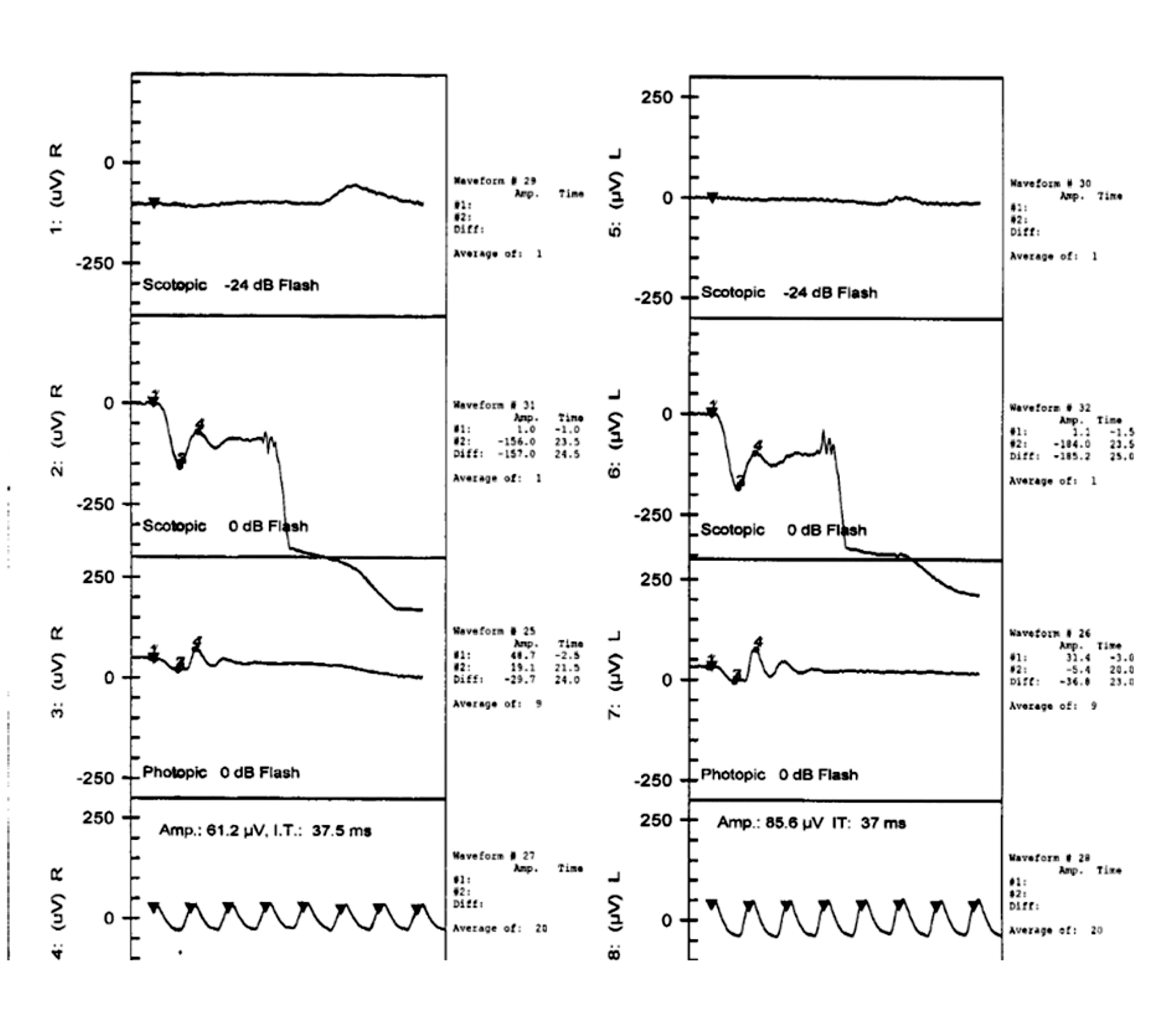
Figure 7: Electroretinogram of the right and left eyes. The scotopic ERG to dim flash is nondetectable and there is an electronegative waveform to standard flash.
Shortly thereafter, the patient noted further worsening of vision in the right eye and was found to have greyish subretinal material consistent with CNV and a small amount of subretinal hemorrhage along the foveal margin of two atrophic lesions in the inferior macula of the right eye (Figures 8A and 8B). Fluorescein angiography revealed late leakage from the CNV in the right eye (Figures 9A and 9B). Spectral domain OCT and angiography showed subretinal fluid and a CNV complex in the right macula (Figure 10A and 10B). Intravitreal bevacizumab (0.125mg/0.05ml) induced regression of CNV in the right eye, with BCVA improving to 20/40.
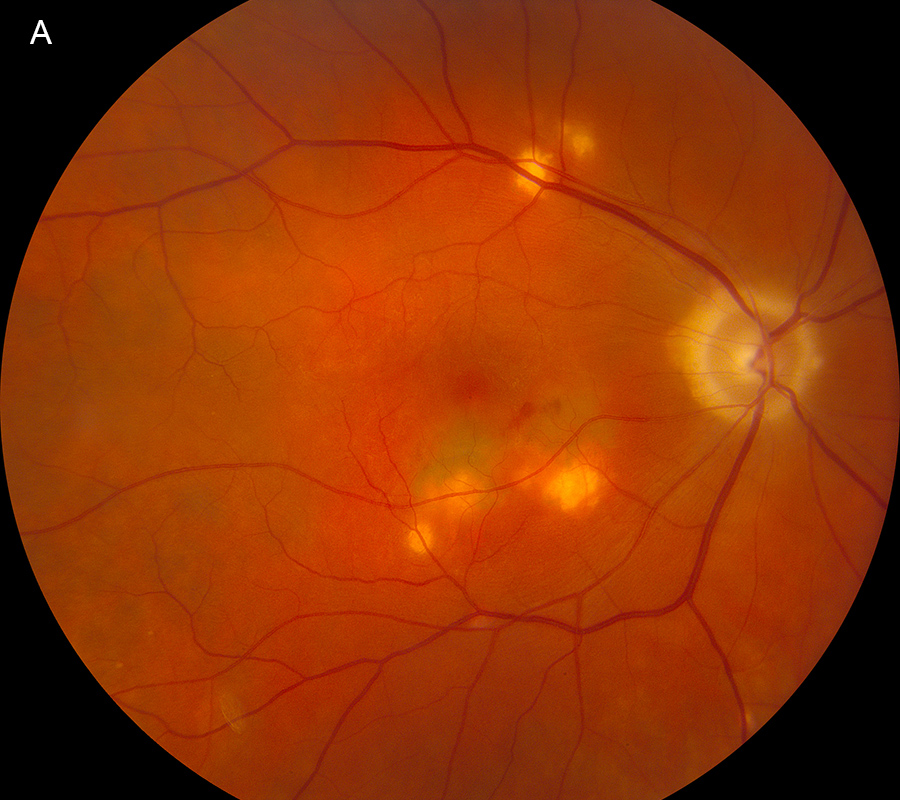
Figure 8A: Color photograph of the right eye. Several new areas of chorioretinal scarring are present. Subretinal hemorrhage and a 'dirty-grey' appearance is seen in the right macula.
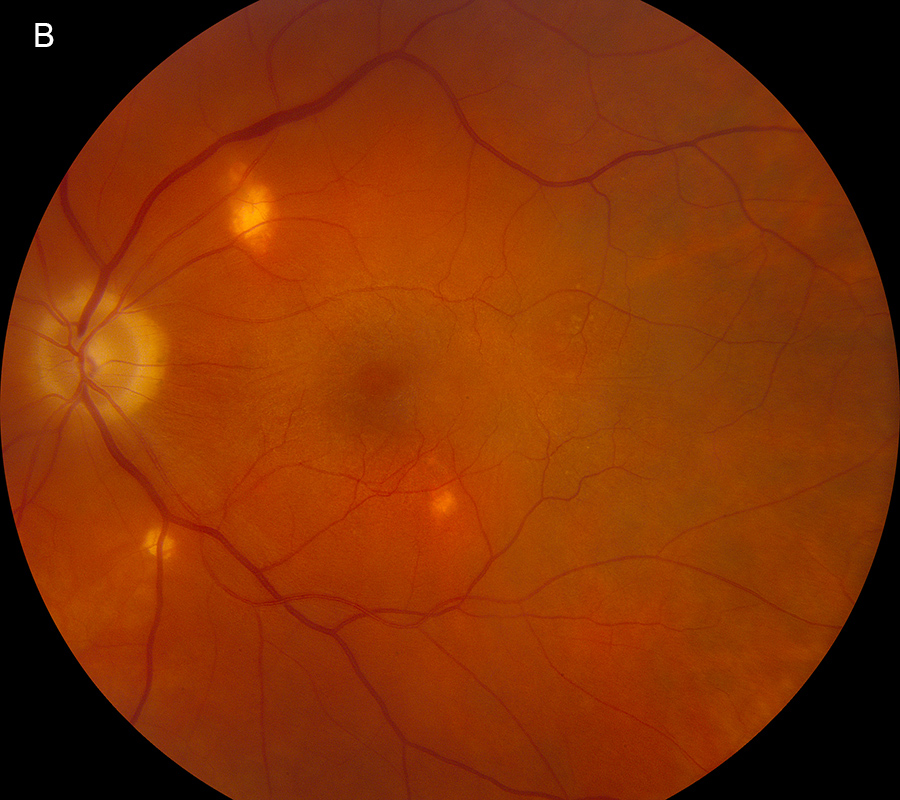
Figure 8A: Color photograph of the left eye. Several new areas of chorioretinal scarring are present.
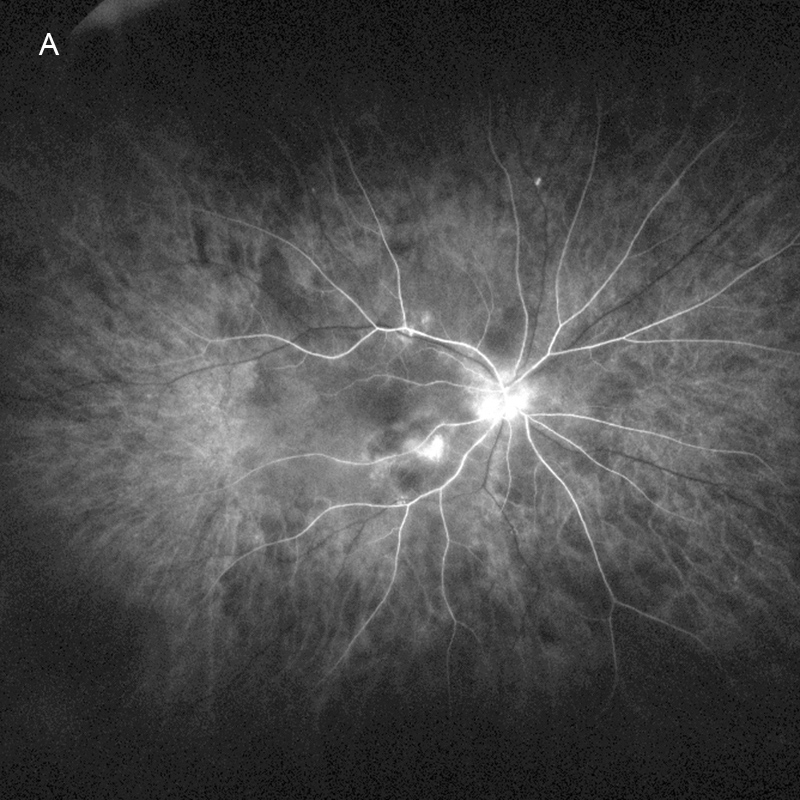
Figure 9A: Fluorescein angiogram of the right eye. The areas of scarring are hyperfluorescent.
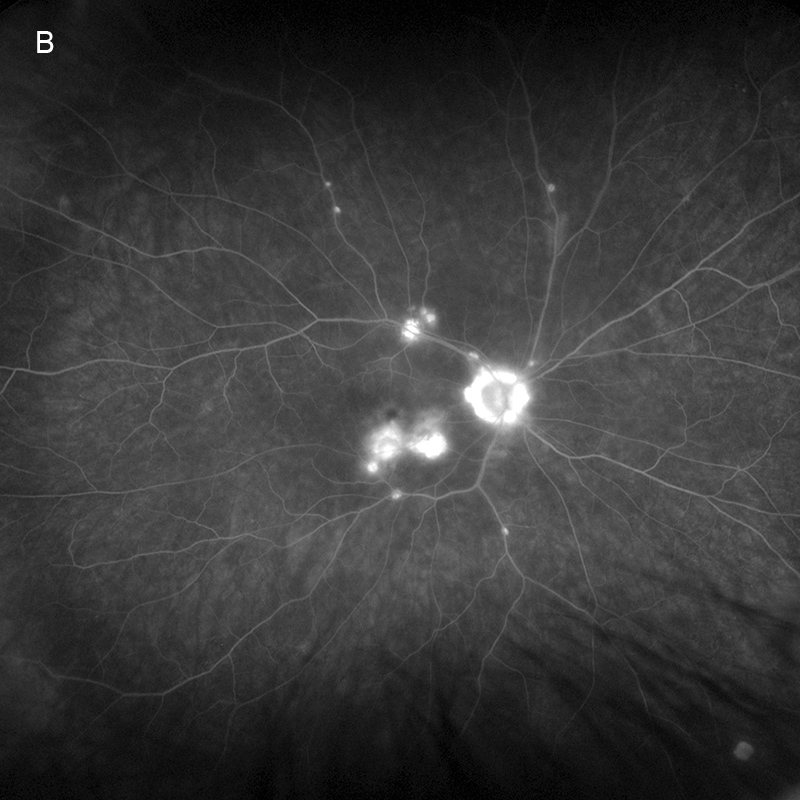
Figure 9A: Fluorescein angiogram of the right eye, late phase. There is significant leakage in the macula and staining of the chorioretinal scars.
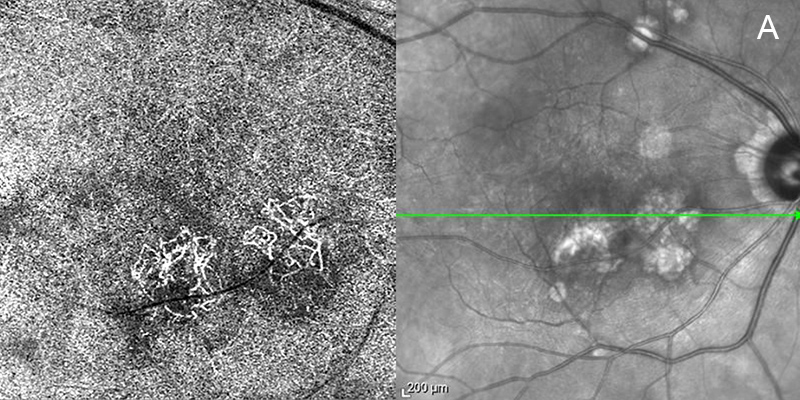
Figure 10A: Spectral domain OCT angiogram and infrared image of the right eye. Note the flow signal on the angiogram corresponding to choroidal neovascularization.
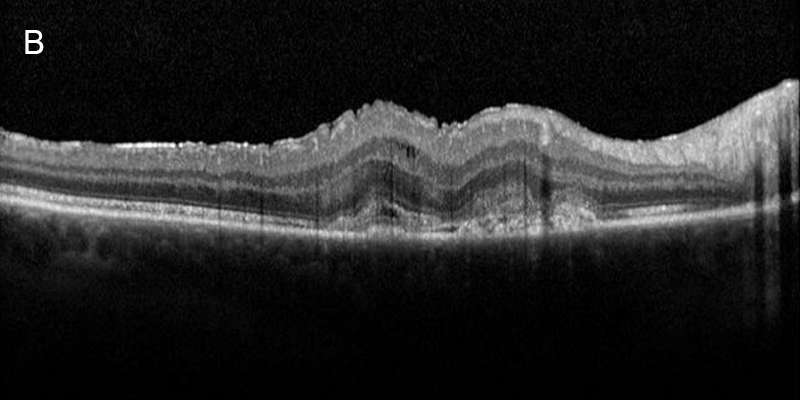
Figure 10B: Spectral domain OCT of the right eye. Subretinal fluid, is present and a shallow RPE elevation secondary to type 2 choroidal neovasculariaation.
Discussion
Paraneoplastic retinopathies have been reported in patients with metastatic melanoma and can be divided into three distinct clinical presentations: MAR, paraneoplastic vitelliform retinopathy, and VKH-like uveitis.1-5 At presentation, patients with MAR may have a normal fundus appearance. With time, however, retinal vascular attenuation, optic nerve pallor, vitreous cells, and RPE disruption may develop.1 All patients with MAR either have extinguished or electronegative waveform activity on ERG, and autoantibodies against retinal bipolar cells are frequently detected.1,6 More recently, bilateral vitelliform-like lesions have been described in patients with metastatic melanoma.2 Typical fundus findings include multifocal, yellow-orange vitelliform lesions associated with subretinal fluid, RPE detachments, and/or deposits of hyper-reflective material involving the outer retinal layers or overlying the RPE on OCT (see Jan, 2011 Case of the Month).2 Various autoantibodies have been detected to support a paraneoplastic etiology, most notably those directed against retinal bipolar cells.2 Electroretinographic results have most commonly been normal, but reports of mild reductions in a- and/or b-wave amplitudes and even electronegative patterns have been reported.2 The least commonly reported ocular paraneoplastic presentation in patients with metastatic melanoma is a VKH-like uveitis, which has only been described three times. In 1978, Sober and Haynes3 described spontaneous bilateral uveitis, poliosis, hypomelanosis, and alopecia in a patient with metastatic cutaneous melanoma. In 1984, Gass4 described a second patient with a history of excised cutaneous melanoma three years prior to presentation who subsequently developed progressive loss of vision to light perception in both eyes, progressive vitiligo of the skin of the face and arms, and deafness over a two-week span. This patient was found to have bilateral panuveitis with what was described as focal choroidal depigmentation and extinguished ERG responses. Lumbar puncture revealed pleocytosis of the cerebrospinal fluid. Biopsy of inguinal lymphadenopathy revealed metastatic melanoma, but full-body CT was otherwise unremarkable. Gass published this case prior to the recognition of MAR, and the extinguished ERG suggested the simultaneous onset of both a VKH-like uveitis and MAR. Most recently, Aisenbrey et al5 described a patient with a history of excised cutaneous melanoma without detectable lymph node involvement who later developed alopecia, poliosis, vitiligo, bilateral panuveitis, optic disc edema, macular edema, and localized choroidal depigmentation and RPE atrophy.
It is noteworthy that our patients also had a diffuse rash that was found on biopsy to be consistent with dermatomyositis, yet another known systemic paraneoplastic complication of metastatic melanoma.7 Other reported non-ocular paraneoplastic manifestations of metastatic melanoma include development of diffuse cutaneous melanosis with dark urine, halo nevi, cachexia, hyperparathyroidism, Cushing’s syndrome from tumor secretion of adrenocorticotropic hormone, disseminated intravascular coagulation, “hot spleen” phenomenon or reversal of the liver-spleen ratio as seen on single photon emission computer tomography, and eosinophilia.7 None of these were evident in our patient, however.
The onset of a VKH-like uveitis may be a favorable prognostic indicator for longer survival in patients with metastatic melanoma. Vitiligo, a clinical finding often associated with VKH, has been associated with longer than expected survival in patients with metastatic melanoma and thought to be a reflection of the body’s successful immunologic suppression of the metastatic melanoma cells.3,8 Patients with metastatic melanoma treated with checkpoint inhibitors have also been reported to develop a VKH-like uveitis,9 again supporting the notion that this particular paraneoplastic presentation may represent a robust host immune response against the malignant cells. Aisenbrey et al5 postulated that long-term use of methotrexate to treat VKH-like disease in their patient may have contributed to the development of metastatic melanoma, which was diagnosed five years after initiation of immunomodulatory therapy and ten years after initial diagnosis of cutaneous melanoma. All other reports of VKH-like uveitis were described in patients who were either known to have, or were concurrently diagnosed with, metastatic melanoma. The patient in our report was treated with long-term oral corticosteroid therapy and remains without signs of recurrent malignancy six years following presentation. Three of the four reported patients with metastatic melanoma and VKH-like uveitis have survived longer than expected for their diagnosis. The one patient who expired 15 months following diagnosis of metastatic melanoma appeared to possess both MAR and VKH-like uveitis at the time of presentation.
In addition to a VKH-like uveitis, patients who have received treatment with checkpoint inhibitor class medications for metastatic melanoma have also been reported to develop focal RPE or chorioretinal atrophy with or without subsequent CNV formation.10 The pathogenesis of the chorioretinal atrophy and subsequent CNV in this patient likely resulted from a similarly potent immune response directed against melanocytes. Other reports have shared our observation that the resultant CNV responds well to intravitreal injections of bevacizumab or ranibizumab, with resolution of subretinal fluid and regression or stabilization of the CNV.10
In summary, paraneoplastic ocular manifestations of metastatic cutaneous melanoma may present in three distinct forms: MAR, vitelliform retinopathy, or VKH-like uveitis. In addition, patients who present with VKH-like uveitis may develop later complications of focal chorioretinal atrophy with or without CNV. Our patient appears to have been the first to develop several sequential and distinct ocular paraneoplastic complications of metastatic cutaneous melanoma, including VKH-like uveitis with focal chorioretinal atrophy, MAR, and secondary CNV. Those few reported patients who did develop VKH-like uveitis following metastatic cutaneous melanoma tended to show better than expected overall outcomes.
Adapted from: Ng CC, Alsberge JB, Qian Y, Freund KB, Cunningham ET Jr. Vogt-Koyanagi-Harada-like Uveitis Followed by Melanoma-Associated Retinopathy with Focal Chorioretinal Atrophy and Choroidal Neovascularization in a Patient with Metastatic Cutaneous Melanoma. Retinal Cases & Brief Reports. 2020 Dec;Publish Ahead of Print. DOI: 10.1097/icb.0000000000001113.